WHO WE ARE

Primary Access and Navigation: The Foundation of Procedural Success The Primary Access and Navigation portfolio integrates Neimor’s micron-precision engineering to secure seamless entry into complex urological pathways. By synchronizing the ApexFlow™ Guidewire series with UroPath™ Introducer Needles, DynaCore™ Ureteral Catheters, and Vestra™ Sheaths, we deliver a validated system designed for high-torque control and atraumatic navigation. This mechanical synergy ensures uncompromising predictability from initial puncture to target localization.
Read More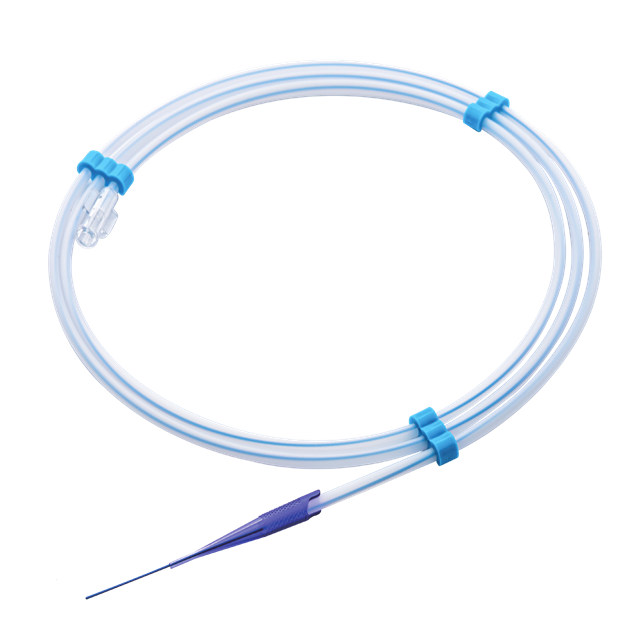
Stone Management and Interventional Therapy: Precision in Clearance The Stone Management and Interventional Therapy portfolio is engineered for efficient stone clearance and stable post-procedural recovery. By integrating the Zenith™ Retrieval Basket with DynaCore™ J Stents and Drainage Catheters, Neimor provides a synchronized system for atraumatic navigation and reliable fluid management. This validated clinical pathway optimizes luminal integrity and procedural safety during complex lithotripsy and renal drainage. Neo for More Precise.
Read More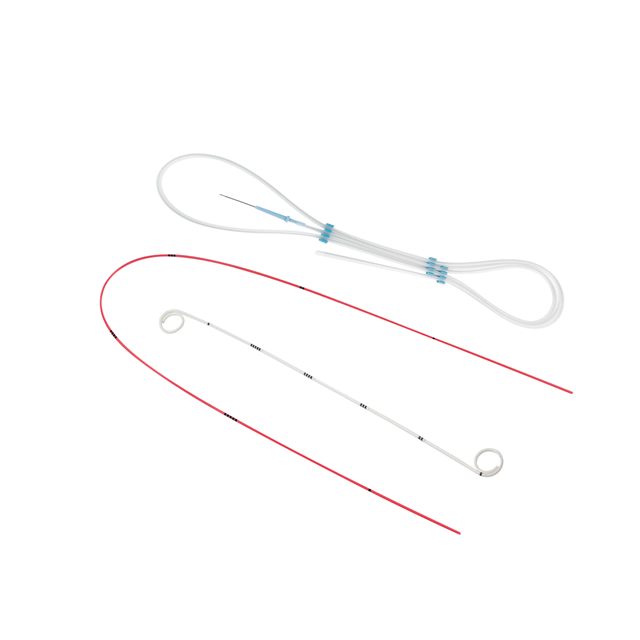
UroPath™: Integrated Solutions for Percutaneous Excellence The UroPath™ Series synchronizes the surgical pathway from initial puncture to definitive drainage. Engineered for mechanical synergy, this validated portfolio eliminates fragmentation in PCNL and Suprapubic access. By integrating precision-matched components, UroPath™ mitigates procedural variability, ensuring uncompromising efficiency and superior patient safety across every complex interventional case.
Read More
VascClamp™ Andrology Solutions: Specialized in advanced circumcision technology. The VascClamp™ series—available in Medical-grade Plastic and Precision Titanium Staple models—delivers superior aesthetic outcomes with minimal bleeding. Our Automatic Displacement Technology ensures painless staple release, supporting the Fast-Track Surgery (ERAS) protocols for modern andrological clinics.
Read More
UroVise™: Redefining Diagnostic Precision in Urodynamicsynamics The UroVise™ Series integrates Neimor’s micron-precision extrusion with advanced sensor compatibility to capture real-time pressure dynamics. Available in both Water-Filled and Air-Charged configurations, our catheters are engineered for superior baseline stability and minimal signal interference. By synchronizing high-fidelity data acquisition with biocompatible polymer architecture, UroVise™ provides the clinical clarity required for accurate bladder function assessment and perioperative risk management. Neo for More Precise: Engineering insight into every diagnosis.
Read More
DiaCore™: Precision Engineered Dialysis Solutions DiaCore™ optimizes extracorporeal therapy through micron-precision extrusion and biocompatible polymers. Our validated Disposable Substitution Lines (Types A–F) ensure universal connectivity and superior luminal uniformity, reducing shear stress. Experience enhanced procedural predictability with Neimor’s audit-ready, GMP-certified dialysis management system. Neo for More Precise: Engineering life-sustaining excellence.
Read More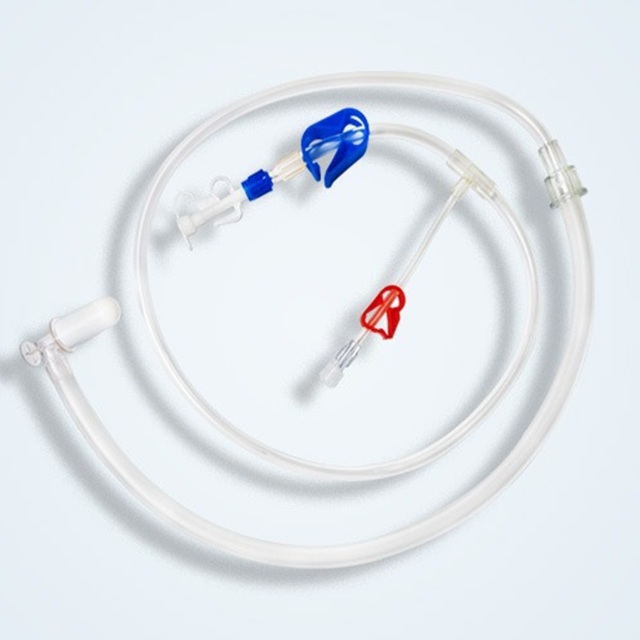
Neimor Custom Manufacturing (OEM/ODM): Leveraging our 2,200m² GMP-certified infrastructure, Neimor Medical provides end-to-end Contract Manufacturing services for global medical device innovators. We specialize in the high-precision fabrication of urological catheters, nitinol components, and complex polymer assemblies. From custom extrusion profiles to micro-precision wire grinding, our vertical integration ensures cost-efficiency and uncompromising quality. Whether you require private-labeling for our existing portfolio or bespoke engineering for new surgical instruments, Neimor is your trusted partner for bringing Class II and III medical devices from concept to market-ready excellence.
Read More
Advanced production lines with Class 100,000 clean rooms, ensuring product quality meets international standards.
Read moreISO 13485 certified quality management system, with strict quality control at every production stage.
Read moreDedicated R&D team continuously improving product design and developing new urology surgery solutions.
Read morePrecision Manufacturing. Clinical Excellence. Changzhou Neimor Medical Technology Co., Ltd. is a leading ISO-certified manufacturer of Class II and III medical polymer devices, driven by the core mission: “Neo for More Precise.” Strategically headquartered in the West Taihu Medical Industrial Park, we bridge the gap between advanced material science and surgical excellence, focusing on specialized interventions in Urology, Nephrology, and Minimally Invasive Surgery. Our production backbone is anchored by a 2,200m² state-of-the-art GMP facility, featuring integrated Class 10,000 and Class 100,000 cleanrooms. We leverage advanced precision catheter extrusion and deep-processing lines to transform complex clinical requirements into high-performance medical realities. From our flagship DynaCore™ ureteral stents and UroNavi™ hydrophilic guidewires to the VascClamp™ circumcision stapler series, every product embodies our commitment to micron-level accuracy. Neimor Medical is more than a supplier; we are a professional solutions provider and OEM/ODM partner dedicated to elevating global healthcare standards. By integrating a computerized quality management system with a steadfast commitment to user trust, we deliver the precision surgeons demand and the safety patients deserve.
🛡️When it comes to treating large or complex kidney stones, modern urology has moved far beyond the era of large incisions. Today, the magic happens through a tiny "tunnel" created by a Single-Use Dilatation Set. If you’ve been told you…